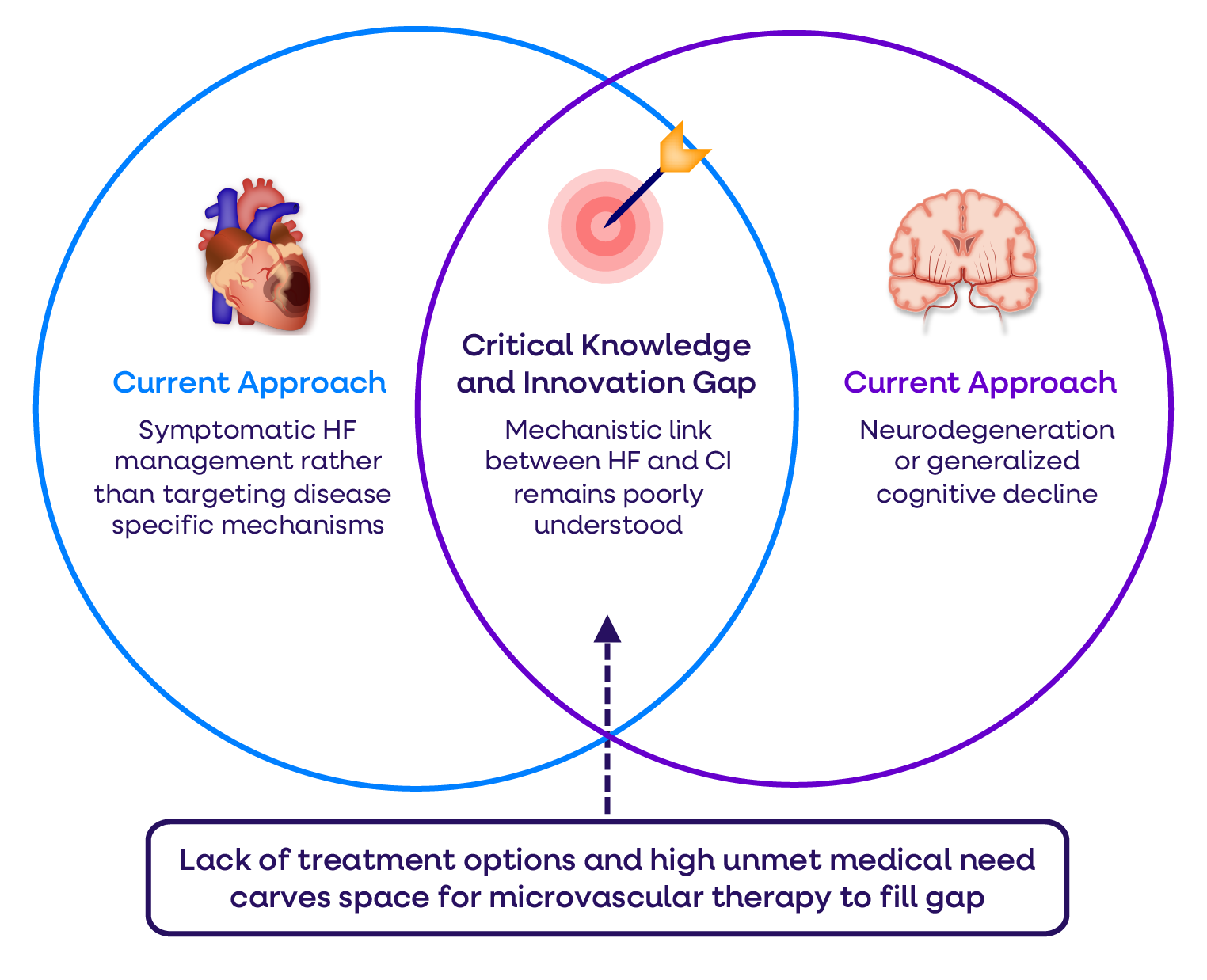
‣ Improved cognitive function leading to better quality of life for HF patients
‣ Enhanced treatment adherence, resulting in improved clinical outcomes and reduced mortality
‣ Improved self-care behaviors, reducing healthcare utilization andassociated costs
Repositioning an existing cystic fibrosis drug with favourable safety profile
Short 30-day treatment period with active vs. placebo arms
Cerebral MRI scans and cognitive tests pre- and post-treatment
60 stable heart failure patients with reduced ejection fraction (HFrEF ≤40) on standard-of-care
Participants recruited by their circle of care team in Toronto